

Ion Exchange Softener
Softener Water Treatment Process
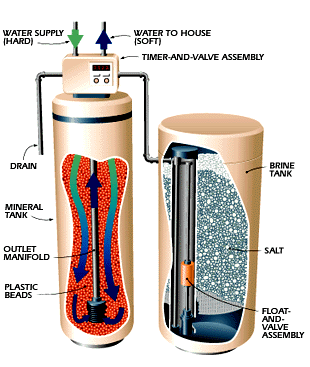
The other method commonly used for water softening is ion exchange softening, also known as zeolite softening. shows below the ion exchange softener. Ion exchange softening exchanges calcium and magnesium ions in water for sodium ions as the hard water passes through a softener. The softener is similar in design to a pressure filter, with resins in place of the filter media.
During treatment, water enters the softener and is directed by a baffle. The water passes through a bed of resin underlain by a bed of gravel, and then is collected by an under-drain and piped out of the softener.
The resins are insoluble solids with attached cations or anions capable of reversible exchange with mobile ions of the opposite sign in the solutions, which pass through them. There are many types of resins, which can be used in ion exchange units. One type of resin commonly used in water softening is zeolite resin, In the case of the ion exchange softening; sodium ions are attached to the insoluble solids of the resins. When water passes through the softener, the sodium ions are exchanged for calcium and magnesium ions in the water. The calcium and magnesium ions are retained on the resin grains. The water leaving the softener has sodium ions in the place of calcium and magnesium ions in its compounds.
Since sodium ions do not cause hardness, the treated water is no longer hard. In a properly operating softener, the treated water will have a zero hardness. To prevent corrosion due to excessively soft water, some quantity of the source water is blended with the softened water downstream of the softener to ensure that the product water has permissible hardness. Ion exchange softening is effective at removing both carbonate and non carbonate hardness and is often used for waters high in non carbonate hardness and with a total hardness less than 350 mg/L.
Disadvantages of Ion – exchange softening are:
*However, ion exchange softening has its disadvantages as well. The calciumand magnesium in the hard water are replaced by sodium ions, which may cause problems for certain people with health problems.
* Softeners have to be backwash in a manner similar to a water filter, and the recharge water, known as brine, can cause disposal problems.
Softener Regeneration
After backwash, the softener is ready to be regenerated. This is the part of the process in which the magnesium and calcium ions on the resin become replaced with sodium so that the softener can be used to treat more hard water. In order to regenerate the resin, a salt solution, known as brine, is allowed to flow through the softener for about an hour.
When dissolved in water, the salt dissociates into its constituent ions – Na+ and CI: The sodium ions replace the calcium and magnesium ions on the resin in the following manner (where “R” preceding an ion means that the ion is bound to the resin.
RCa + NaCl → RNa + CaCl2
RMg + NaCl → RNa + MgCl2
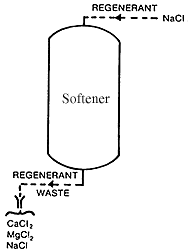
- Regeneration of Resins in Softener
During regeneration, calcium chloride, magnesium chloride, and excess sodium chloride flow to waste. The Above figure shows the regeneration process in ion exchange softener. After the brine has been given a sufficient contact time, it must be rinsed out of the softener. During the rinse cycle, fresh water is passed through the unit as it would be during treatment, but with the effluent going to waste. Rinse usually takes about 20 to 40 minutes. Both the spent brine from regeneration and from the rinse must be disposed of carefully since the calcium, magnesium, and sodium salts are corrosive and toxic to the environment. Spent brine is sometimes discharged in sewers or into streams at very high dilutions. Alternatively, the brine can be disposed of in a landfill.
3 Responses to “Ion Exchange Softener”
Leave a Reply







 LIKE TO GET UPDATES
LIKE TO GET UPDATES  TO GET EXPERT GUIDE
TO GET EXPERT GUIDE
Can you provide more information on this? take care
We are looking for install water softener plant at Industrial area, jodhpur
Sir,
I have 30 years experience in Water treatment worked as HOD in 235mw power plant.So please appoint me me as distributor.
With regards,
R.Prabhakar Rao
Mob.7780704483
Email: prabhakar.3691@gmail.com